
A Fluid Solution to Dendrite Growth in Batteries
A new paper from associate professor Jiandi Wan’s group published in Science Advances proposes a potential solution to dendrite growth in rechargeable lithium metal batteries using microfluidics. In the paper, the group proved that flowing ions near the cathode can potentially expand the safety and lifespans of these next-generation rechargeable batteries.
“My students and I are quite excited to be published in one of the Science journals,” said Wan. “It means a lot to know that such a broad research community is interested in our work.”
Lithium (Li) metal batteries are a type of Li battery that uses Li metal as the anode. These batteries have a high charge density and potentially double the energy of conventional Li ion batteries, but safety is a big concern. When they charge, some ions are reduced to Li metal at the cathode surface and form irregular, tree-like microstructures known as dendrite, which can eventually cause a short circuit or even an explosion.
The theory is that dendrite growth is caused by the competition of mass transfer and reduction rate of Li ions near the cathode surface. When the reduction rate of ions is much faster than the mass transfer, it creates an electroneutral gap called the space-charged layer near the cathode that contains no ions. The instability of this layer is thought to cause dendrite growth, so reducing or eliminating it might reduce dendrite growth and therefore extend the life of a battery.
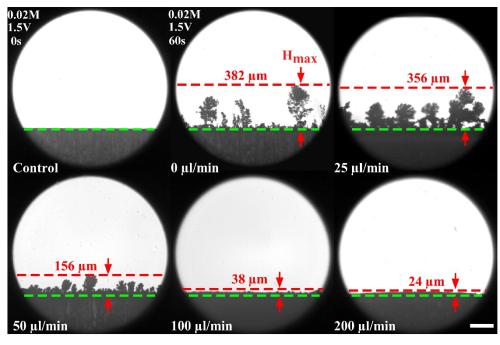
Wan’s idea was to do this by flowing ions through the cathode in a microfluidic channel to restore a charge and offset this gap. In the paper, team outlined their proof-of concept tests that bringing more ions into the cathode is an effective strategy for reducing dendrite, finding that this flow of ions could reduce dendrite growth by up to 99%.
For Wan, the study is exciting because it shows the effectiveness of applying microfluidics to battery-related problems and paves the way for future research in this area.
“With this fundamental study and microfluidic approaches, we were able to quantitatively understand the effect of flow on dendrite growth,” he said. “Not many groups have studied this yet.”
Though it is likely not possible to directly incorporate microfluidics in real batteries, Wan’s group is looking at alternative ways to apply the fundamental principles from this study and introduce local flows near the cathode surface to compensate cations and eliminate the space charge layer.
“We are quite excited to explore the new applications of our study,” he said. “We are already working on design of the cathode surface to introduce convective flows.”
The paper, titled “Suppression of Dendrite Growth by Cross-Flow in Microfluidics” was published in the February 2021 issue of Science Advances, a sister journal of Science that publishes research from all STEM fields.
